HR+ Breast Cancer: What Is It And What Are Your Options If Diagnosed?

If you’ve been recently diagnosed with HR+ breast cancer or know someone who has, there are probably a lot of things going through your mind: What does my breast cancer diagnosis really mean? How is this specific type of cancer going to affect me? How long is treatment going to last?
We’re here to reduce some of that uncertainty surrounding your diagnosis and help you play a more active part in your breast cancer treatment.
What HR-positive means
HR, short for hormone receptor-positive, is a form of breast cancer that grows in response to estrogen (ER + breast cancer) and/or progesterone (PR+), hormones naturally produced by the body. Approximately 80% of breast cancer cases are ER+, with 65% of those also being PR+.1 If diagnosed, you’re likely to take anti-estrogen therapy for five years to reduce the risk of the cancer returning,2,3 BUT for some women, longer treatment (through year 10) could help.
What to know about the treatment process
Anti-estrogen therapy, aka endocrine therapy, includes medications that affect levels of hormones in two different ways. One common medication works by blocking the action of estrogen on cancer cells in the breasts. A common example of this drug type is called Tamoxifen and is often prescribed for pre-menopausal women. In contrast, aromatase inhibitors, including Arimidex, are meant to stop the body from producing estrogen altogether—and are also typically prescribed for post-menopausal women.
Ask any woman enduring endocrine therapy, and she’ll tell you, being on hormone inhibitors is NO joke. While the treatment can save your life, the many side effects can really take a toll on your body. Plus, in trials studying extended endocrine therapy, only 5% of women benefited from long-term therapy. This means that for the other 95%, longer treatment did not impact the risk of recurrence.2-7
Breast Cancer Index™ (BCI) identifies which women are among those likely to benefit from continued treatment. By providing personalized information, BCI enables peace of mind to stop treatment at year five for those who are not likely to benefit, and the dedication to continue for those who are.
“I could have danced on the ceiling! I was so happy to know that I wasn’t going to have to take this medicine that had really changed my life in ways I didn’t like,” exclaimed Mary Darden, NP and Breast Cancer Index™ patient.
As you approach the five-year mark after your initial diagnosis, you can talk to your doctor about whether the side effects and potential risks outweigh the benefits of an additional five years of treatment.
The Breast Cancer Index™ Test helps personalize decision-making
Breast Cancer Index™ is the only test recognized by the National Comprehensive Care Network® to predict extended anti-estrogen therapy benefits.2 The test provides two pieces of valuable information to help you and your doctor make a personalized decision:
1. Whether or not an additional five years of anti-estrogen therapy is likely to help reduce your risk of cancer returning.
2. Your individual risk of distant (metastatic) recurrence after the first five years of therapy (based on your original tumor biology).
That means avoiding overtreatment for those for whom therapy has no impact beyond five years.3-8 And for patients like Jelina Roger, the test confirmed how vital it was for her to continue taking her medication—she couldn’t even miss a dose or two.
Breaking down the BCI Test process
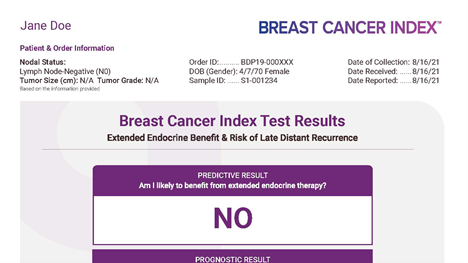 It’s super easy! In fact, you don’t have to do anything beyond asking your oncologist for the test. The test uses the tissue from your original biopsy, and the results will automatically be sent to your doctor in about seven days for them to go over with you. The test results will be in one clear, easy-to-understand report where you get a straightforward ‘yes’ or ‘no’ as to whether extended endocrine therapy will work and your percentage of late distant recurrence risk.
It’s super easy! In fact, you don’t have to do anything beyond asking your oncologist for the test. The test uses the tissue from your original biopsy, and the results will automatically be sent to your doctor in about seven days for them to go over with you. The test results will be in one clear, easy-to-understand report where you get a straightforward ‘yes’ or ‘no’ as to whether extended endocrine therapy will work and your percentage of late distant recurrence risk.
What to know about insurance coverage
Depending on your individual plan, BCI may be covered by your insurance. Medicare also covers BCI at a predetermined rate with no fees charged to you (subject to specific coverage criteria).9 Plus, the test is available through a variety of payment options, including a Financial Assistance Program if you meet the requirements. Once your doctor has ordered the test, a Patient Services team member will be in touch with you to walk you through your options and what to expect regarding the billing process.
Bonus! Download the conversation guide for a printable list of questions to take directly to your doctor and patient brochure to learn more about BCI.
Disclaimer: This article is for informational use only and is not intended for or implied to be a substitute for medical advice. Always seek the guidance of your health care professional for any question you may have regarding medical advice, diagnosis, or treatment. Please contact your medical professional for specific advice regarding your health and treatment. This information may be relevant in the U.S. and other markets and is not intended as a product solicitation or promotion where such activities are prohibited. Because Hologic materials are distributed through websites, eBroadcasts and tradeshows, it is not always possible to control where such materials appear. For specific information on what products may be available in a particular country, please write to womenshealth@hologic.com.
Hologic, 3D, 3D Mammography, Genius, Breast Cancer Index, the Science of Sure, and associated logos are trademarks and/or registered trademarks of Hologic, Inc. and/or its subsidiaries in the United States and/or other countries.
SOM-01190
-
1. How to Read Hormone Receptor Test Results. BreastCancer.org: https://www.breastcancer.org/symptoms/diagnosis/hormone_status/read_results. Accessed December 22, 2021.
2. Referenced with permission from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Breast Cancer V.3.2021. © National Comprehensive Cancer Network, Inc. 2021. All rights reserved. Accessed March 24, 2021. To view the most recent and complete version of the guideline, go online to NCCN.org. NCCN makes no warranties of any kind whatsoever regarding their content, use, or application and disclaims any responsibility for their application or use in any way.
3. Davies C, et al. Lancet. 2013;381:805-816.
4. Gray R, et al. J Clin Oncol. 2013;31:(suppl; abstr 5).
5. Jakesz R, et al. J Natl Cancer Inst. 2007;99:1845-1853.
6. Goss PE, et al. J Natl Cancer Inst. 2005;97:1262-1271.
7. Mamounas EP, et al. NSABP B-42. GS4-01. SABCS 2019.
8. Goss PE, et al. N Engl J Med. 2016;375:209-219.
9. MolDX: Breast Cancer IndexTM (BCI) Gene Expression Test. CMS.gov: https://www.cms.gov/medicare-coverage-database/view/lcd.aspx?LCDId=37822&ContrId=360&ver=7&ContrVer=1&CntrctrSelected=365*1&Cntrctr=365&name=&DocType=4%7C5&LCntrctr=365*1&bc=AAAAAACAIAAA&. Accessed December 10, 2021


